Start of topic | Skip to actions
Numerical Simulations of Gaseous Detonations
Detonations with One-Step Chemistry
Normalization
Under the assumption of a planar stationary detonation wave propagating with constant speed d the inhomogeneous Euler equations can be solved exactly. These planar one-dimensional solutions are used as initial conditions for all simulations with the one-step model. Further on, all following computations for the one-step reaction model utilize dimensionless quantities. With
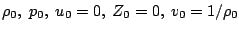
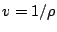
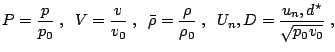
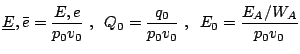
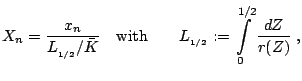
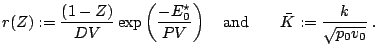
-- RalfDeiterding - 14 Dec 2004